Today we tried to put all the elements together, with a big push on justifying our answers based on the observations.
I alos gave some model observations based on the questions in your Scipad
I alos gave some model observations based on the questions in your Scipad
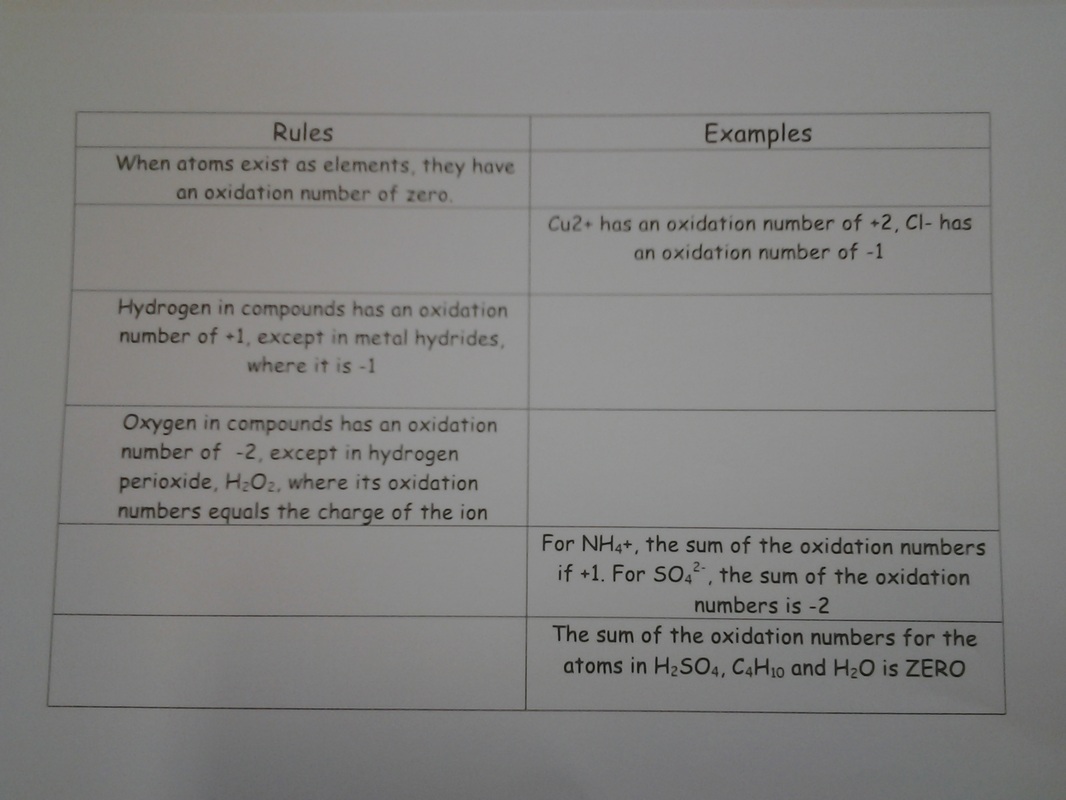
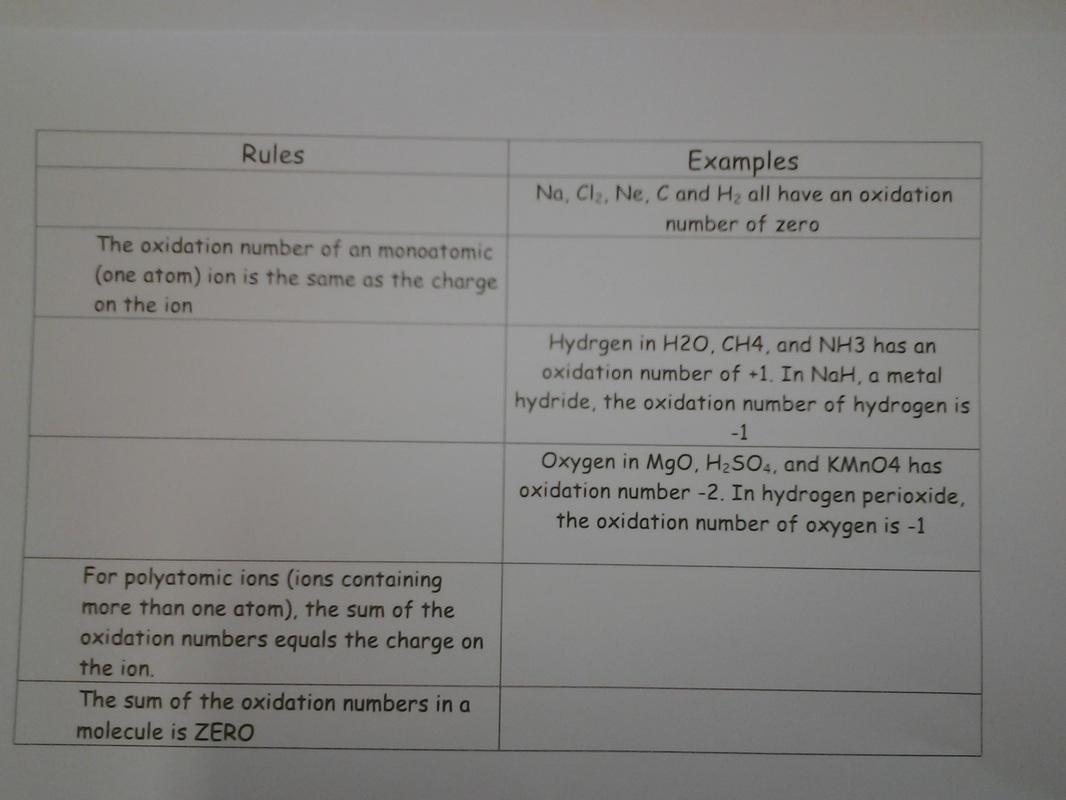
I stressed the fact you need complete explanations for justifying the oxidant and reductant using both the 1/2 equations and oxidation numbers - with the footnote you should state the oxidation numbers of the relevant species.
Another practice question you could do is (from earlier in the week)
1) Rachel gets a piece of Copper metal and places in it a test tube with dilute Nitric acid.
The copper fizzes rapidly and a brown gas is given off. The solution turns a pale blue/green colour. She notices the piece of metal is gone at the end of the reaction.
Or
2mL or acidified potassium dichromate is placed in a test tube. Some sodium hydrogen sulphite is added. The colour changes from orange to green/blue.
Another practice question you could do is (from earlier in the week)
1) Rachel gets a piece of Copper metal and places in it a test tube with dilute Nitric acid.
The copper fizzes rapidly and a brown gas is given off. The solution turns a pale blue/green colour. She notices the piece of metal is gone at the end of the reaction.
Or
2mL or acidified potassium dichromate is placed in a test tube. Some sodium hydrogen sulphite is added. The colour changes from orange to green/blue.



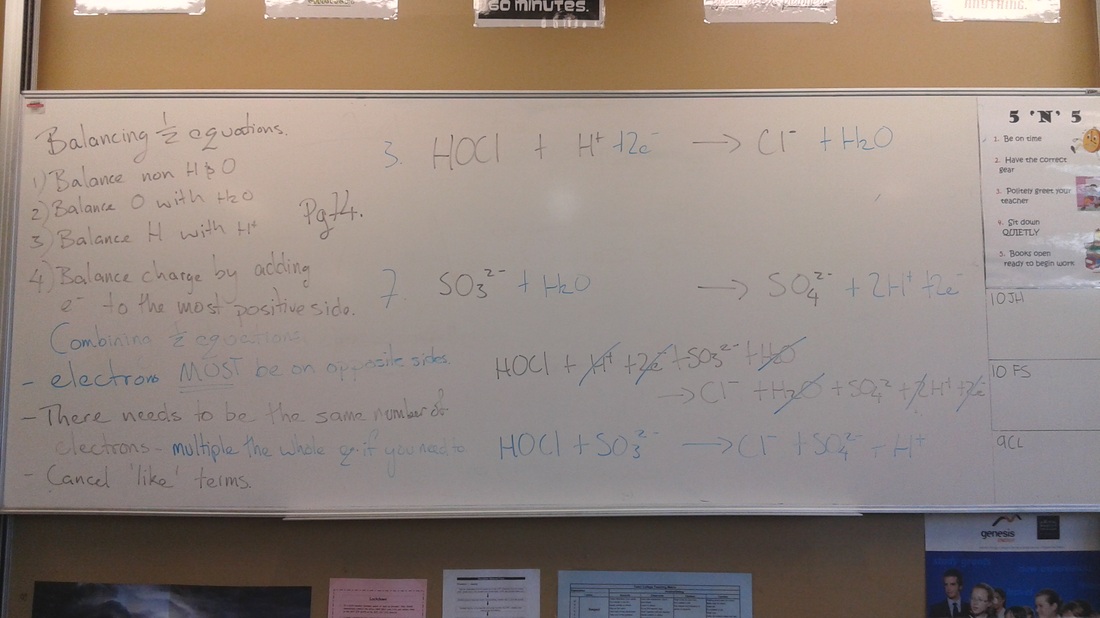











 RSS Feed
RSS Feed
