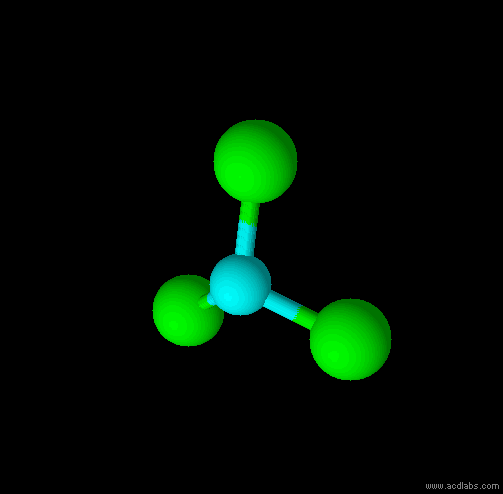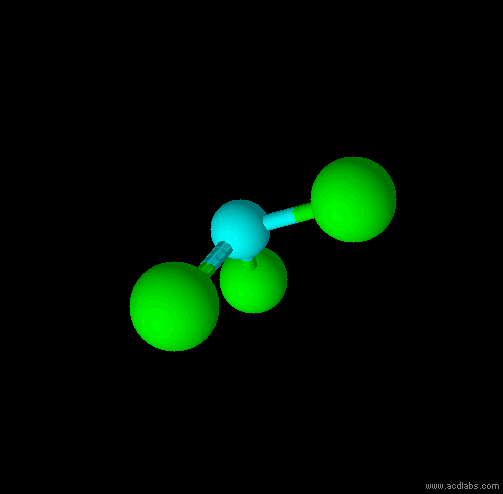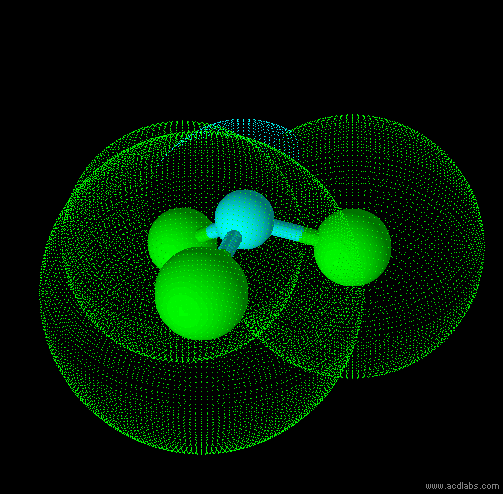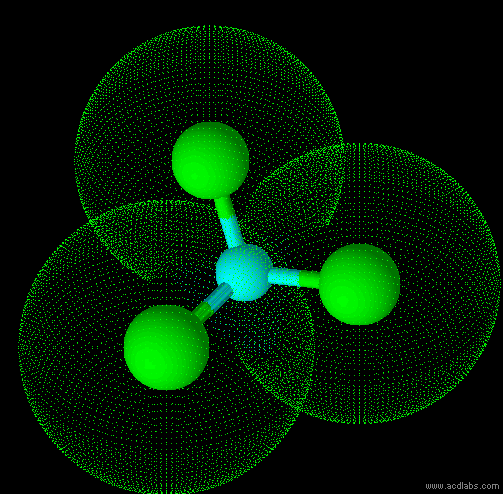Shapes and polarity
Here is a great website for shapes of molecules
Below are the different shapes you need to know for Level 2 Chemistry
Below are the different shapes you need to know for Level 2 Chemistry
|
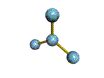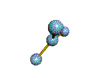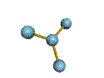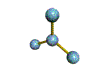
This is a Tetrahedral shape
The are 4 regions of negative charge around the central atom that repel for maximum separation to make a bond angle of 109.5 degrees. Because all 4 regions are bonded, the shape is tetrahedral. This shape is trigonal pyramid. There are 4 regions of negative charge around the central atom that repel for maximum separation to give a bond angle of 109.5 degrees. Because only 3 regions are bonded and 1 region is non-bonded the shape is trigonal pyramid. This shape is triangular plannar There are 3 regions of negative charge around the central atom that repel for maximum separtion to give a bond angle of 120 degrees. Because all 3 regions are bonded the shape is triangular plannar. |