|
So I am thinking the better late than never with the lesson posts :-)
Carboxylic acids are formed when PRIMARY alcohols are oxidised (using the reagent acidified permanganant or acidified dichromate). Carboxylic acids turn blue litmus red, and acts like any other acid when reacted with metal to produce hydrogen gas, or carbon dioxide gas if it reacts with a carbonate. Amines are formed via a substitution reaction from haloalkanes. The reagent is ammonia (NH3). Amines are basic, and will turn red litmus blue. We also did a summary diagram on the board - a big flowchart connecting all the reactions and reagent together. There is also the electronic flow chart on the home page Don't forget to check the ultranet page for the some extra notes and summary sheets too. Woohoo, Mrs C got her laptop back. YAY Which means she will be back to updating the blog Today we revisited Markovnikovs rule - 'the rich get richer', and Saytseff's rule 'the poor get poorer' rules for addition reactions (which can form alcohols) and elimination reactions (which occur to alcohols) In the interim we also covered polymers.... I'll put some notes up tomorrow
today we covered cis and trans isomers.
Remember isomers are organic molecules with the same molecular formula (so the same number of atoms) but a different arrangement in space (so straight chained compared to branched chained etc) The conditions required for cis trans isomers to form is 1) there must be a double bond 2) the carbon atoms either side of the double bond must have 2 different functional groups/groups of atoms attached. We use molymods to good effect to help us see the difference. We then did the correspon Today we did a summary of alkenes, they are very similar to alkanes in their physical properties, although when reacted with oxygen (combustion) they tend to produce a much sootier flame
Today we had a bit of a content dump about haloalkanes and the reactions they undertake. Don't panic if you don't quite get it yet, it will come as we learn about the different organic families
We also watched a video of a gecko trying to climb teflon. Teflon is a polymer made from a halogen called tetraflouro ethene Today we quickly recapped the properties of alkanes, and then went over how to name different structural isomers (as well as an intro into what isomers are). We also briefly talked about different formula types. And then we practised naming alkanes in Scipad. We will move onto reactions of alkanes and the properties of haloalkanes tomorrow.
Today we started on some of the content for the organic content by going over some of the properties of alkanes I also due you attention to the template will be using for learning in this topic -SOLO (structure of observed learning outcomes). This gives a frame work for surface, deep and conceptual understanding and will hopefully help you connect some of the ideas and content we learn about. So look out for the posters that will be coming to B2 soon :-) We also spent some time making a resource to help you learn the prefixes for naming organic compounds - you should do what works best for you. But remember
Meth, Eth, Prop, But, Pent, Hex, Hept, Oct is the same as 1, 2, 3, 4, 5, 6, 7, 8. And then some naming practice, I got round some of you on how to name branched alkanes and we will be picking this up on Monday What better way to start a new topic than doing a quick revision of the old one. So we quickly went through the Atomic structure and bonding topic. We then went through a power point introducing the key concepts of Organic chemistry, and how it is like learning a new language - you need to learn the basics first and then suddenly you can make a sentence, and then a paragraph.
Have fun |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
November 2014
Categories |












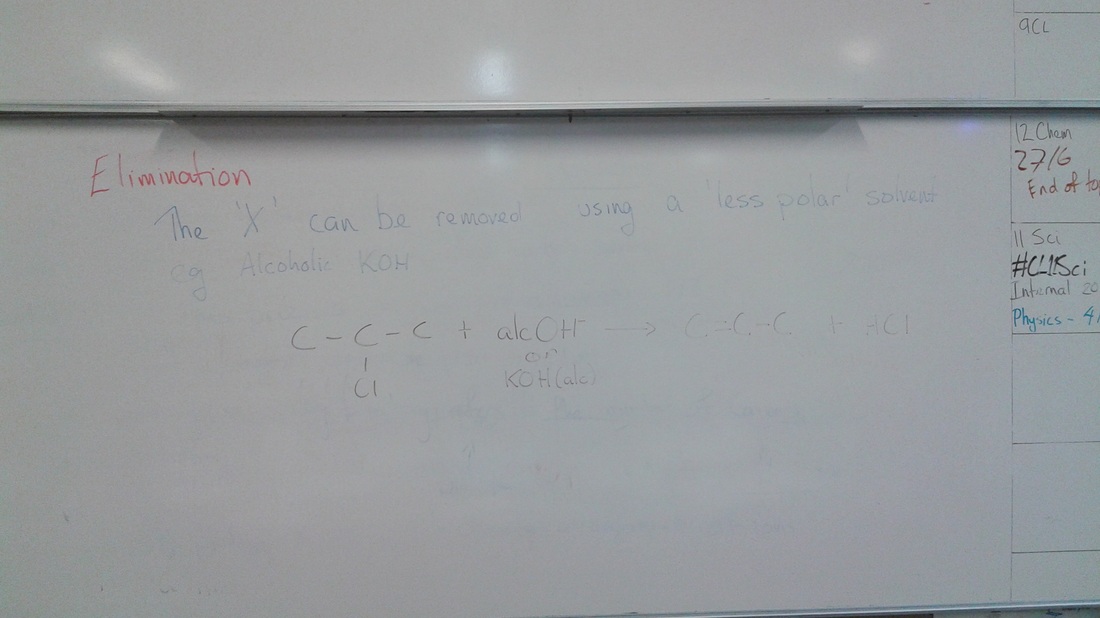








 RSS Feed
RSS Feed
