Today we had a bit of a content dump about haloalkanes and the reactions they undertake. Don't panic if you don't quite get it yet, it will come as we learn about the different organic families
| I stressed that you need to be specific about the reactions and the conditions, for example reacting an alkane with aqueous KOH (so just dissolved in water) with result in a substitution reaction and the formation of alcohol, but alcoholic KOH (so KOH dissolved in alcohol) will result in an elimination reaction and the formation of an alkene. We then were in desperate need of a prac, so we compared the reactions of alkanes (cyclohexane) and alkenes (cyclohexene) with bromine water (Br2) and oxygen (combustion). |
We also watched a video of a gecko trying to climb teflon. Teflon is a polymer made from a halogen called tetraflouro ethene


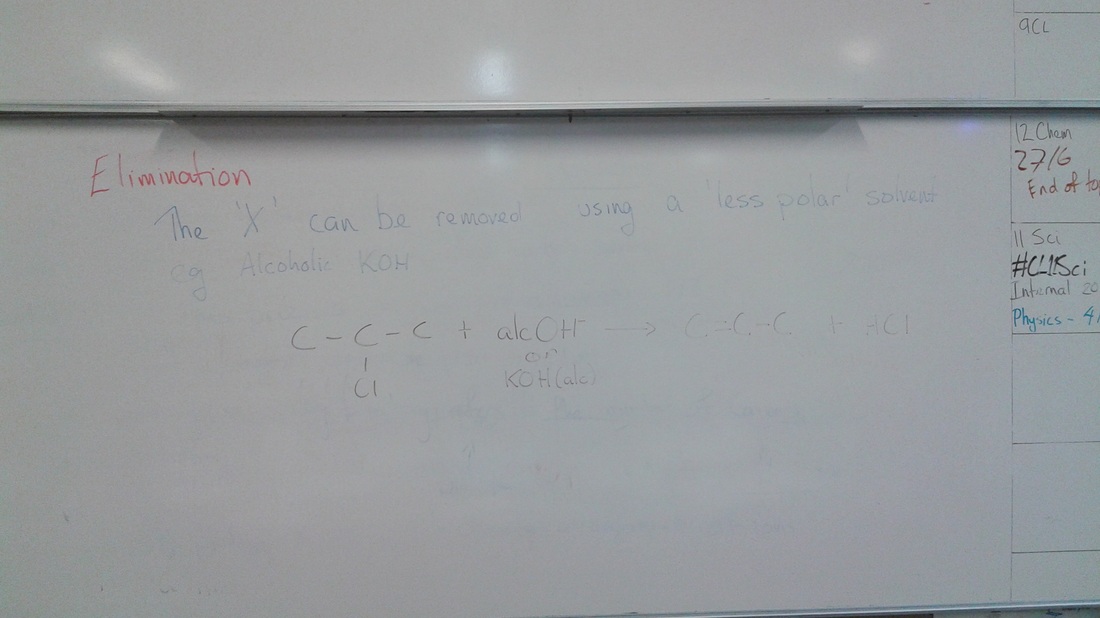


 RSS Feed
RSS Feed
